Author Guidelines
Template File
Title page Template (Original article) Template (Review) Template (Case report)Copyright Transfer Agreement Patient consent form Endnote style
Enacted on December 28, 2022
Enacted on January 30, 2026
1.Overview
The Kawasaki Disease (Kawasaki Dis.; KD; eISSN 2983-3183) is the official journal of the Korean Society of Kawasaki Disease (KSKD). The journal is issued biannually on the last days of June and December. The journal is operated according to the Principles of Transparency and Best Practice in Scholarly Publishing 4th ed (https://publicationethics.org/resources/guidelines-new/principles-transparency-and-best-practice-scholarly-publishing). KD is an open access journal. All articles are distributed under the terms of the Creative Commons Attribution Non-Commercial License (https://creativecommons.org/licenses/by-nc/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
A. Aims and scope
KD is the official, peer-reviewed, open-access journal of the Korean Society of Kawasaki Disease (KSKD). The journal serves as a multidisciplinary platform for disseminating clinical and scientific knowledge related to Kawasaki disease and Kawasaki-like syndromes, with particular emphasis on advancing understanding, diagnosis, treatment, and outcomes.
The journal targets a broad international audience, including pediatricians, pediatric cardiologists, infectious disease specialists, thoracic surgeons, radiologists, and other healthcare professionals and researchers involved in the care of patients with Kawasaki disease or related vasculitides.
KD publishes a wide range of article types, including original research articles, clinical and translational reviews, case reports, editorials, and other scholarly works approved by the Publications Committee. Topics may include clinical management, basic science, imaging techniques, surgical innovations, epidemiology, and long-term sequelae of Kawasaki disease.
B. Article Processing Charge
There are currently no article processing charges (APCs) for submissions to this journal. Should there be any changes regarding APC policy, updated information will be provided on the journal’s website.
C. Editorial policy
The editorial board presumes that all authors listed in a manuscript have agreed to the following policy of KD on submission of the manuscript.
Any authors may submit manuscripts of original articles, reviews, editorials, case reports, and special contributions written in Korean or English. Except for negotiated secondary publications, manuscripts submitted to the journal must be previously unpublished and not under consideration for publication elsewhere.
The editorial board reserves the right to make corrections, both literary and technical, to the papers. Under any circumstances, the identities of the reviewers will not be revealed. It is the author's responsibility to ensure that a patient's anonymity should be carefully protected, to verify that any experimental investigation with human participants reported in the manuscript was performed with informed consent, and to follow all guidelines for experimental investigation with human participants required by the institution(s) with which all the authors are affiliated.
If an author should be added or deleted after the submission, it is the responsibility of the corresponding author to ensure that the authors concerned are aware of and agree to the change in authorship. The editorial board assumes no responsibility for disputes regarding authorship changes.
KD is published biannually on the last day of June and December. All published articles become permanent properties of the KSKD and may not be published elsewhere without written permission.
D. Online Access
Full-text files of KD are freely available on the journal homepage. (https://www.e-kd.org)
E. Contact us
KD Editorial Office
The Korean Society of Kawasaki Disease
33, Sinbanpo-ro 23-gil, Seocho-gu, Seoul, Apt. 1403, Building 202, Republic of Korea
Tel : +82-2-3010-3924
E-mail: e-kd@kawasaki.or.kr
2. Copyright and License
A. Copyright
All published articles become permanent intellectual property of the KSKD and may not be republished elsewhere without written permission. Copyrights of the articles are owned by the KSKD. Each author must sign the authorship responsibility and copyright transfer agreement, attesting to authorship criteria. The corresponding author submits the Copyright Transfer Form during submission. Authors must obtain and provide written permission for any previously published material. Submitted material will not be returned unless requested.
B. Open access
KD is an open-access journal. All articles are distributed under the terms of the Creative Commons Attribution Non-Commercial License (https://creativecommons.org/licenses/by-nc/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
3. Research and Publication Ethics
Regarding policies on research and publication ethics not addressed in these instructions, authors should refer to the Committee on Publication Ethics (COPE) guidelines on good publication (https://publicationethics.org/guidance), the Recommendations for the conduct, reporting, editing, and publication of scholarly work in medical journals by the ICMJE (https://www.icmje.org/recommendations/) or Good Publication Practice Guidelines for Medical Journals (KAMJE, https://www.kamje.or.kr/guide/books).
A. Statement of human and animal rights and informed consent
Any investigations involving humans and animals should be approved by the institutional review board (IRB) or institutional animal care and use committee (IACUC), respectively, of the institution where the study took place. In addition, investigations with pathogens requiring a high degree of biosafety should obtain approval from the relevant committee (institutional biosafety committee). Informed consent should be obtained, unless waived by the IRB, from patients (or legal guardians) who participated in clinical investigations. Human participants should not be identifiable, such that patients' names, initials, hospital numbers, dates of birth, or other protected healthcare information should not be disclosed. If experiments involve animals, the research should be based on national or institutional guidelines for animal care and use. Original articles submitted to KD that address any investigation involving humans and animals should include a description of whether the study was conducted under an approval by the IRB (with or without patient informed consent) or IACUC, respectively. IRB approval number is required for the submission process, and if absent, the process cannot proceed. When appropriate, the editorial office of KD may request official documentation of IRB or IACUC approval. All human studies are expected to follow the ethical principles outlined in the Declaration of Helsinki (https://www.wma.net/policies-post/wma-declaration-of-helsinki/).
B. Authorship and author’s responsibility
Authors must meet all aspects of the following 4 criteria:
(1) Substantial contributions to the conception or design of the work; or the acquisition, analysis or interpretation of data;
(2) Drafting the work or revising it critically for important intellectual content;
(3) Final approval of the version to be published;
(4) Agreement to be accountable for all aspects of the work and ensuring that questions related to the accuracy or integrity are appropriately investigated and resolved.
Anyone who does not meet the above criteria may be listed as a contributor in the Acknowledgments section.
Only one corresponding author is allowed. A footnote indicating that two first authors contributed equally is permissible, as long as the authors certify equal first author role. The editorial board does not allow adding or changing authorship (including first or corresponding authors) after the manuscript has been accepted. Any change in the byline (such as addition or deletion of authors, or changing the order of names) requires a signed letter from all authors indicating their agreement. The editorial board assumes no responsibility for such changes.
The corresponding author takes primary responsibility for communication with the journal during the manuscript submission, peer review, and publication. They typically ensure that all the journal’s administrative requirements, such as providing authorship details, ethics committee approval, clinical trial registration documentation, and conflicts of interest (COI) forms and statements, are properly completed, although these duties may be delegated to one or more coauthors. The corresponding author should be available throughout the submission and peer review to respond to editorial queries. They should be available to respond to critiques and cooperate with journal requests for data, additional information, or questions about the paper even after publication. Authors may appeal the editorial decisions via e-mail (e-kd@kawasaki.or.kr). Appeals are processed according to procedures set by COPE (https://publicationethics.org/resources/discussion-documents/authorship).
C. Originality and duplicate publication
Manuscripts under review or published by other journals will not be accepted for publication in KD, and articles published in this journal are not allowed to be reproduced in whole or in part in any form of publication without the permission of the editorial board. Figures and tables can be used freely if the original source is properly cited in accordance with the Creative Commons Non-Commercial License. It is mandatory for all authors to resolve any copyright issues when citing a figure or table from a different journal that is not open access.
A similarity Check is used to screen manuscripts for plagiarism or duplicate publication. Plagiarism is defined as reproducing another’s work without attribution. If plagiarism is detected before or after acceptance, or after publication, the author will have the opportunity for rebuttal. Unsatisfactory rebuttals will result in retraction and a publication ban for a period determined by the editorial board.
D. Secondary publication
Manuscripts may be republished if they meet the ICMJE conditions for secondary publication as follows: certain types of articles, such as guidelines issued by governmental agencies and professional organizations, may need to reach the widest possible audience. In such instances, editors sometimes intentionally publish material that is simultaneously published in other journals, with the consent of the authors and the editors of those journals. Secondary publication, in the same or another language, especially in other countries, is acceptable and may be beneficial if the following conditions are met. The authors must obtain approval from the editors of both journals (the editor concerned with secondary publication must have a photocopy, reprint, or manuscript of the original article). The primary publication must be given precedence, with a minimum interval of one week before the secondary publication, unless a different arrangement is agreed upon by both editors.
The secondary publication should target a different audience; an abbreviated version may be appropriate. The secondary version must accurately reflect the data and interpretations of the primary publication. The footnote on the title page of the secondary version informs readers, peers, and documenting agencies that the paper has been published in whole or in part and cite the original publication. A suitable footnote might read: “This article is based on a study first published in [Journal Title], with full citation.”
E. Conflicts of interest
A COI may exist when an author, or the author’s institution or employer, has financial or personal relationships or affiliations that could bias the author’s decisions regarding the manuscript. All authors must disclose any relevant financial interests or financial conflicts, including those that existed during the conduct of the research and at the time of publication. This includes future financial interests such as patent applications that may result in potential personal gain. All disclosures of any potential COI, including specific financial interests and relationships and affiliations (other than those affiliations listed in the title page of the manuscript) relevant to the subject of their manuscript will be disclosed by the corresponding author on behalf of each coauthor, if any, as part of the submission process. Likewise, authors without COI will be requested to state so as part of the submission process. If authors are uncertain about what constitutes a relevant financial interest or relationship, they should contact the editorial board. Failure to disclose COI will result in rejection or withdrawal of the manuscript. For all accepted manuscripts, each author’s disclosures of COI, relevant financial interests and affiliations, and declarations of no such interests will be published. The policy requesting disclosure of COI applies for all manuscript submissions. If an author’s disclosure of potential COI is determined to be inaccurate or incomplete after publication, an erratum will be published to rectify the original published disclosure statement. Authors are also required to report detailed information regarding all financial and material support for the research and work, including but not limited to grant support, funding sources, and provision of equipment and supplies as part of the submission process. For all accepted manuscripts, each author’s source of funding will be published.
The authors should disclose all potential COI. If there is a disclosure, the editors, reviewers, and readers can interpret the manuscripts with this understanding.
If a co-author is in a personal relationship with another (e.g., a person younger than 19 years, a spouse or a first-degree cousin), this should be marked in the byline with related facts written in a footnote. If necessary, the editorial board may request consent to provide personal information from the corresponding author, or an investigation from the affiliated institution. If a research misconduct by co-authors in a personal relationship is confirmed, the related fact can be notified to the relevant institution (e.g., advancement, employment, promotion, and research funding order, etc.) from which the author took advantage.
Submission by Editors
Final decisions regarding manuscript publication are made by the editor-in-chief or a designated editor who does not have any relevant conflicts of interest. In the event that an editor has a conflict of interest with a submitted manuscript or with the authors, the manuscript will be handled by one of the other editors who does not have a conflict with the review and who is not at the same institution as the submitting editor. In such circumstances, full masking of the process will be ensured so that the anonymity of the peer reviewers is maintained.
F. Process to manage research and publication misconduct
When the journal faces suspected cases of research and publication misconduct such as redundant (duplicate) publication, plagiarism, fraudulent or fabricated data, changes in authorship, a fraudulent undisclosed conflict of interest, ethical problems with a submitted manuscript, a reviewer who has appropriated an author’s idea or data, and complaints against editors, the resolution process will follow the guidances provided by COPE (https://publicationethics.org). The discussion and decision on the suspected cases are carried out by the Editorial Board and Research Ethics Council.
G. Editorial responsibilities
The editorial board is committed to the ongoing monitoring and protection of publication ethics. This includes: establishing clear guidelines for article retraction; plagiarism screening for all manuscripts (https://www.ithenticate.com/); maintaining the integrity of the academic record; preclusion of business needs from compromising intellectual and ethical standards; publishing errata, clarifications, retractions and apologies when necessary; preventing plagiarism and fraudulent data. The responsibilities of the editorial board include: having the authority to accept or reject articles; avoiding conflicts of interest regarding decision; accepting a manuscript when reasonably certain of its validity; promoting the publication of correction or retraction when errors are identified; and preserving the anonymity of reviewers.
4. Publishing Policies
A. Data sharing
The journal encourages authors to state the data sharing in their submission. Authors may state linking to a repository or declaring confidentiality of the data. Since 2019, all manuscripts reporting clinical trials must be submitted with a data sharing statement (Table 1). If authors describe this in their manuscripts, the description will be published alongside their manuscripts.
Table 1. Examples of data sharing statements that fulfill the ICMJE requirements
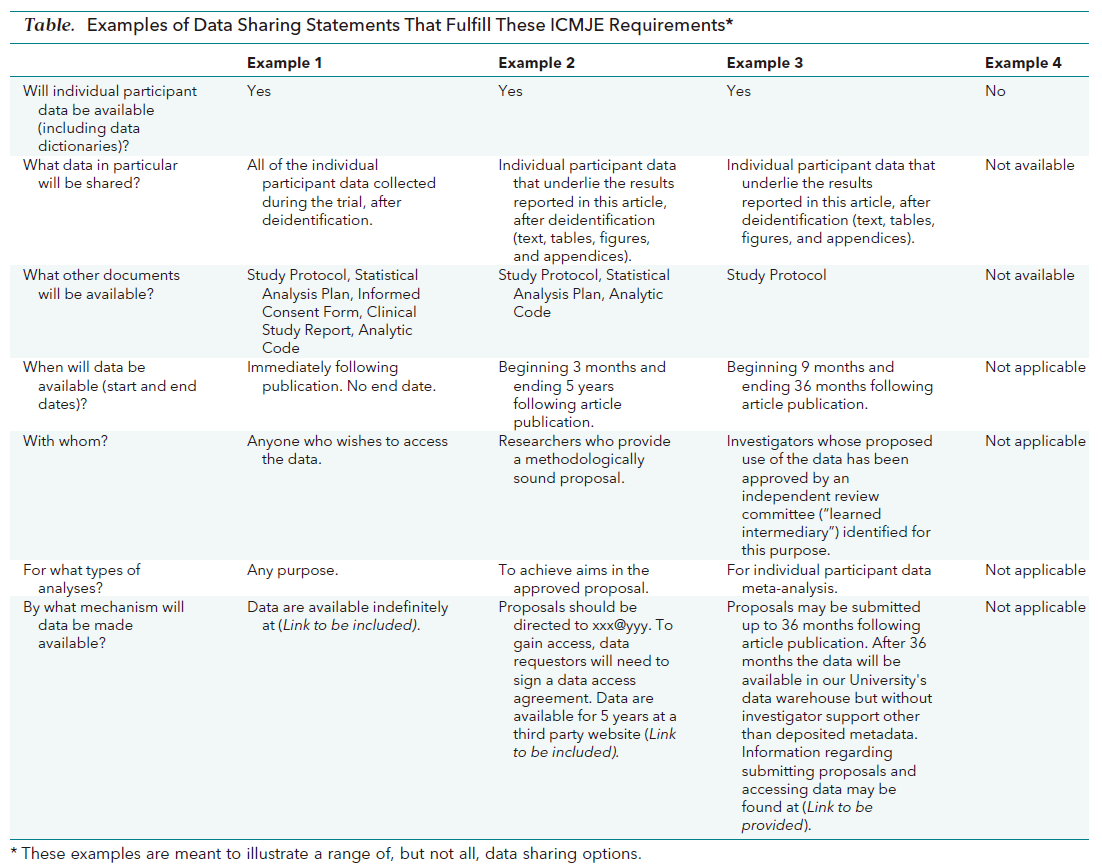
B. Preprint
KD allows the submission of manuscripts that have previously been posted on preprint servers. In such cases, authors should indicate the preprint server and DOI in the cover letter. Once the article is published in KD, the preprint should be linked to the final published KD version via its DOI (e.g., “This article has been published in Kawasaki Disease following peer review and can be viewed at [DOI].”), and the KD version should be cited, instead of the preprint. Citation of preprints is not permitted.
C. Article sharing
KD is an open access journal under the terms of the Creative Commons Attribution Non-Commercial License (http://creativecommons.org/licenses/by-nc/4.0/) where articles can be shared without embargo. Articles can be shared only as an accepted or published version, as described below.
Accepted versionThe accepted version incorporates all amendments made during peer review but precedes the final published version. The accepted version may be placed on:
- The author's personal website
- The author's company/institutional repository or archive
- Non-profit repositories preprint servers or repositories
- Directly provided to students or research collaborators for personal use
It is recommended that the authors include a note and DOI link on the first page.
Published versionThe final published version can be shared immediately upon publication under the Creative Commons Attribution Non-Commercial License, permitting unrestricted non-commercial use, distribution, and reproduction in any medium, provided the original work is properly cited.
D. Archiving
KD provides the electronic backup and preservation of access to the journal content in the event the journal is no longer published by archiving in National Library of Korea (https://www.nl.go.kr/archive/search_eng.do?op=all&kwd=2983-3183).
E. Use of Artificial Intelligence (AI)
Authors may use generative AI and AI-assisted technologies (e.g., ChatGPT) to improve the clarity and language quality of their manuscripts. However, such tools must be employed with appropriate human oversight to ensure the accuracy and reliability of the output. Authors are responsible for carefully reviewing and editing any content produced by AI, as it may appear plausible but still contain errors. AI tools and technologies must not be credited as authors or cited as such, as authorship entails accountability and intellectual contributions that can only be assumed by humans. If generative AI was used during manuscript preparation, the name of the specific tool and the extent of its use must be clearly disclosed in the Methods or Acknowledgments section. This policy does not apply to the routine use of basic tools for grammar, spelling, reference checks, and similar editorial functions. Please note that authors are ultimately accountable for all aspects of the submitted work, including any use of AI or AI-assisted technologies. Adherence to this policy ensures that AI is used in a transparent, responsible, and ethically sound manner, consistent with best practices in scientific publishing.
5. Preparing manuscripts
A. Categories of manuscripts
KD publishes original article, review, editorial, case report, and special contribution.
Original article: Original investigations in areas relevant to Kawasaki Disease. Maximum 4,000 words, 6 tables or figures, and 35 references. Include a structured abstract and its length should not exceed 300 words. Additional material may be placed in appendices.
In case of Korean article, less than 8 pages of the printed journal are recommended for original article. (1 page of this journal contains about 2,500 Korean characters when only characters are included in the page). Original articles should adhere to the relevant reporting guidelines for their study design, as listed by the EQUATOR Network (e.g., STROBE for observational studies, CONSORT for randomized trials).
Systematic review: Systematic reviews are critical assessments and evaluations of research (not simply summaries) that attempt to address a focused clinical question using methods designed to reduce the likelihood of bias. Meta-analyses combine this with aggregate analyses. This kind of manuscripts should be structured into the subheadings: introduction, main subject, and conclusion. Such articles must be compliant with relevant guidelines and include a narrative abstract and its length should not exceed 300 words. In case of systematic review, the length is limited to 5,000-8,000 words with a maximum of 100 references. All systematic review submissions are required to follow the PRISMA (Preferred Reporting Items for Systematic Reviews and Meta-Analyses) guidelines.
Review: Clinical reviews should address specific clinical questions or relevant issues regarding Kawasaki disease. Such articles should summarize current research relevant to the questions they address, be evidence-based to the extent possible, be balanced, and detail the importance of the questions or issues. Include a narrative abstract and its length should not exceed 300 words. Do not combine a case report with your review. This kind of manuscript should be structured into subheadings: introduction, main subject, and conclusion. The length is limited to 5,000 words. The number of figures, tables, or references are not limited.
Editorials: It is written upon the request of the editorial board. It covers authoritative comments or opinions on controversial matters with significant implications for Kawasaki disease, or provides in-depth analysis and criticism of articles published in the journal. Maximum 1,500 words.
Case report: Case report must be a brief description of a previously undocumented disease process, a unique manifestation or treatment of a known disease process or unreported complications of treatment regimens. Entities previously reported elsewhere must be extremely important to be considered. Include a narrative abstract with keywords, introduction, case, discussion focusing on the implications of the case reported, references, and figures or tables. Maximum 1,500 words, 20 references, and 4 tables or figures. The abstract should not exceed 250 words. Case reports must be prepared in accordance with the CARE guidelines.
Special contribution: This kind of manuscript does not conform to a traditional format. Please discuss with the editorial board before submission.
Authors should discuss with the editorial board if their article exceeds the maximum number of words, figures or tables allowed.
Table 2. Recommended maximums for articles submitted to the KD
| Category | Abstract: word count/format | Main: word count | Figures/tables | References |
|---|---|---|---|---|
| Original article | 300/structured | 4,000 | 6 | 35 |
| Review | 300/narrative | 5,000 | NA | NA |
| Editorial | NA | 1,500 | NA | NA |
| Case report | 250/narrative | 1,500 | 4 | 20 |
| Special contribution | NA | NA | NA | NA |
B. Reporting guidelines for specific study designs
For the specific study design, such as randomized control studies, studies of diagnostic accuracy, meta-analyses, observational studies, and non-randomized studies, it is recommended that the authors follow the reporting guidelines listed in Table 3.
Table 3. Reporting guidelines
| Initiative | Type of study | Source |
|---|---|---|
| CONSORT | Randomized controlled trials | http://www.consort-statement.org |
| STARD | Studies of diagnostic accuracy | https://www.equator-network.org/reporting-guidelines/stard/ |
| PRISMA | Preferred reporting items of systematic reviews and meta-analyses | http://www.prisma-statement.org |
| STROBE | Observational studies in epidemiology | http://www.strobe-statement.org |
| MOOSE | Meta-analyses of observational studies in epidemiology | https://www.equator-network.org/reporting-guidelines/meta-analysis-of-observational-studies-in-epidemiology-a-proposal-for-reporting-meta-analysis-of-observational-studies-in-epidemiology-moose-group//a> |
| CARE | Case reports | https://www.equator-network.org/reporting-guidelines/care/ |
| ARRIVE | Animal studies | https://www.equator-network.org/reporting-guidelines/improving-bioscience-research-reporting-the-arrive-guidelines-for-reporting-animal-research/ |
C. Registration of clinical trial research
Any research that deals with a clinical trial should be registered to the primary registry, such as the WHO International Clinical Trials Portal (https://www.who.int/clinical-trials-registry-platform), NIH ClinicalTrials.gov (https://www.clinicaltrials.gov/), ISRCTN Resister (https://www.isrctn.com/), and Clinical research information system (https://cris.nih.go.kr/cris/index.jsp). The clinical trial registration number should be published at the end of the abstract.
D. Preparing manuscripts
1. We accept manuscripts written in English or Korean. Even for Korean manuscripts, the title page should be written in both English and Korean, and the abstract with keywords, references, tables, figures, and figure legends must be written in English. Authors who are non-native speakers should receive English-editing service. All text files should be in Microsoft Word format (DOC or DOCX, 2007 and above), and all figures need to be in JPG/JPEG/TIFF/PPT/PDF format. . The manuscript should be typed in a 12-point font and double-spaced on A4 with a 2cm margin on both sides and a 3cm margin on the top and bottom. Page numbers should start from 1, indicated at the bottom center of each page, beginning with the title page of the submitted manuscript. Line numbers should be displayed on the left side of the manuscript and should continue consecutively without starting anew on each page. We recommend to use the manuscript template.
2. The manuscript should be organized in the following order: full title page (including all the authors’ details) as a separate file; blinded main document in a single file, which starts with abstract with keywords, introduction, methods, results, discussion, acknowledgements (optional), references, and figure legends. Tables and figures should be uploaded separately.
3. The use of abbreviations should be minimized. When used, they should be fully described at first appearance in the text and described in the parenthesis. Use of Korean terms need to be consistent with the latest version of lexicon published from Korean Medical Association (http://term.kma.org). If Korean terms are unavailable (medical terms, proper nouns, place names, drug names or units), authors may use English terms. Even if Korean terms are poorly understandable, authors should use only Korean terms after describing the corresponding English terms in parentheses at first appearance. Leave blanks between English words and subsequent parentheses (c.f., no blanks between Korean words and parentheses).
4. The use of SI units is encouraged. All data should be expressed in metric units. Temperature should be expressed in degrees Celsius Leave blanks in front of units except percent (%) and angle (°). P values should be typed in capital letters ("P") and should be marked as "P value" in the table.
5. KD performs double-blinded review of the submitted manuscripts. The authors' names, affiliations, and any other remarks that may identify the authors should not appear in the main document, figures, and appendices for the blinded review. In case identifying details are found, the editorial board will ask the corresponding author to re-upload the files after deleting such details before referring peer review.
6. Please also refer to KD article templates and recent articles for style.
E. Title page
Include the following items on the full title page.
1. Type of manuscript (e.g., original article)
2. Title: Both English (≤50 words) and Korean (≤50 letters) titles are needed (English manuscripts do not need Korean titles).
3. Full names, affiliations, and order of all authors: Each author’s full name must be provided in the order of first name, middle name, and last name.Academic degrees (e.g. MD, PhD) are not required. When authors are affiliated with different institutions, indicate each affiliation clearly using superscript numbers. For example:
e.g., Mi Young Han1
1Department of Pediatrics, College of Medicine, Kyung Hee University, Seoul, Korea
4. Information of the corresponding author (name, affiliation, ORCID, postal address, phone number, and e-mail address).
5. Running title: This title will be printed at the top of each page of the published paper and should be no longer than 10 words (including spaces). Even Korean manuscripts need English running titles.
6. ORCID, COI, funding sources, and data sharing statements: All authors write their ORCID and clearly disclose their COI and funding sources. A data sharing statement should also be provided. If not applicable, indicate so.
7. Author contributions: Specify each author's contribution according to the CRediT. (Table 4)
Table 4. CRediT statements
Term |
Definition |
|---|---|
Conceptualization |
Ideas; formulation or evolution of overarching research goals and aims |
Methodology |
Development or design of methodology; creation of models |
Software |
Programming, software development; designing computer programs; implementation of the computer code and supporting algorithms; testing of existing code components |
Validation |
Verification, whether as a part of the activity or separate, of the overall replication/ reproducibility of results/experiments and other research outputs |
Formal analysis |
Application of statistical, mathematical, computational, or other formal techniques to analyze or synthesize study data |
Investigation analysis |
Conducting a research and investigation process, specifically performing the experiments, or data/evidence collection |
Resources analysis |
Provision of study materials, reagents, materials, patients, laboratory samples, animals, instrumentation, computing resources, or other analysis tools |
Data Curation analysis |
Management activities to annotate (produce metadata), scrub data and maintain research data (including software code, where it is necessary for interpreting the data itself) for initial use and later reuse |
Writing - Original Draft |
Preparation, creation and/or presentation of the published work, specifically writing the initial draft (including substantive translation) |
Writing - Review & Editing |
Preparation, creation and/or presentation of the published work by those from the original research group, specifically critical review, commentary or revision – including pre-or post-publication stages |
Visualization |
Preparation, creation and/or presentation of the published work, specifically visualization/ data presentation |
Supervision |
Oversight and leadership responsibility for the research activity planning and execution, including mentorship external to the core team |
Project administration |
Management and coordination responsibility for the research activity planning and execution |
Funding acquisition administration |
Acquisition of the financial support for the project leading to this publication |
Example:
Conceptualization: GD Hong.
Data curation: MY Han.
Formal analysis: MY Han.
Methodology: MY Han., GD Hong.
Software: MY Han.
Validation: JH Han.
Investigation: GD Hong.
Writing-original draft: MY Han.
Writing-review and editing: all authors.
8. Acknowledgments (optional): Persons who have made substantial contributions, but who are not eligible as authors are named in the acknowledgments.
9. IRB number: IRB approval number should be typed (e.g., IRB no. ##-##-###).
10. Word count: For English articles, please provide the word count of the main text and figure legends, excluding the title page, abstract, tables, and references.
F. Main body
The main body is a blinded document for review and should contain the following components in a single file, which starts with abstract and keywords, introduction, methods, results, discussion, references, and figure legends. Tables and figures should not be embedded in the text.
Abstract
Abstracts for original articles should be 300 words or less, and structured into the subheadings: Background, Methods (include information on design, setting, participants, interventions, and main outcomes measured), Results, and Conclusion. But narrative abstracts are necessary in review, and case reports. In your results, you should emphasize the magnitude of findings over test statistics, ideally including the size of the effect and its confidence intervals for the principal outcomes. Citations should not be used in the abstract. Abbreviations should be minimized and, if used, must be defined within the abstract by the full term followed by its abbreviation in parentheses. Keywords should be listed using 3-6 Medical Subject Headings (MeSH, https://meshb.nlm.nih.gov/search) joined by semicolons. In case of an RCT, the trial registry number should appear after the conclusion (e.g., NCT01234567).
Main body
1) Introduction
The most effective introduction section should concisely argue how the topic is new, scientifically important, and clinically relevant. Usually, we recommend three paragraphs: The first paragraph to describe the circumstances or historical context that led you to investigate the issue. The second to describe why your investigation is consequential. What are its potential implications? How does it relate to issues raised in the first paragraph? Why is this specific investigation the next logical step? The last to explain the goals of this investigation: Clearly state the specific research objective or hypothesis and your primary outcome measure.
2) Methods
- This section should include subsections with contents that detail the study design, study setting and population, study protocol, measurements or key outcome measures, and data analysis (include sample size determinations and other relevant information, the names of statistical tests, and software used).
- The role of funding organizations and sponsors in the conduct and reporting of the study should be included here.
- When equipment is used in a study, provide in parentheses the model number, name of the manufacturer.
- If citing an in-press paper for the description of methods, please upload a copy of the in-press paper for the editor and reviewers. This in-press material will be handled with appropriate confidentiality.
- Research involving human participants or animals must meet local, legal and institutional requirements, and generally accepted ethical principles such as those set out in the Nuremberg Code, the Belmont Report, or the Declaration of Helsinki.
- Manuscripts reporting data involving human participants must indicate a positive review by an IRB or equivalent. This requirement includes studies that qualify for IRB expedited status. Most institutions require IRB review of studies that qualify for exempt status and require that this determination be made by the IRB, not by the authors. The "Methods" section of the manuscript must explicitly state that IRB approval was obtained, that the IRB determined the study was exempt, or that the study did not involve human participants (e.g., publicly available and previously de-identified information from national data sets, or other studies not meeting the definition of human participants research, additional information available at www.hhs.gov/ohrp/policy/cdebiol.html). IRB approval number should be typed after this remark (e.g., IRB no. ##-##-###). The "Methods" section should also indicate the type of consent used (written, verbal, or waived), and confirm that consent was obtained from all participants (unless waived by the IRB).
- Manuscripts reporting the results of investigations of live vertebrate animals must indicate approval by an IACUC or equivalent. We reserve the right to request submission of IRB or IACUC documentation at any time.
- When working with administrative databases, authors should be diligent in checking the validity of variables (e.g., by cross-checking with other variables in the dataset) and patterns of missing data. Both of these factors can bias results. Authors should also recognize that causal inferences are generally limited when interpreting results from administrative data sources. For analyses using probability samples, care should be taken to use clusters, strata and weights in analyses and that substantially restricting such samples (e.g., to small age groups) may create bias and unusual associations between variables.
3) Results
This section should be concise, and include the statistical analysis. Results presented in tables or figures should be referred to in the text, but the material should not be presented again. In addition to the data collected in the study, the results should also indicate the success of protocol implementation (e.g., was blinding successful, was there a high inter-rater reliability?). In keeping with the recommendations of the Institute of Medicine regarding gender-specific research, ensure correct use of the terms sex (when reporting biological factors) and gender (identity, psychosocial or cultural factors). We ask that "all papers reporting the outcomes of clinical trials report on men and women separately unless a trial is of a sex-specific condition (such as endometrial or prostatic cancer)." (Institute of Medicine (US) Committee on Women's Health Research. Women’s Health Research: Progress, Pitfalls, and Promise. National Academies Press (US); 2010. https://doi.org/10.17226/12908.) If the study was done involving an exclusive population, for example in only one sex, authors should justify the reasons, except in obvious cases.
4) Discussion
Briefly summarize the results and how they relate to your area of investigation. Consider only those published articles directly relevant to interpreting your results and placing them in context. Do not stress statistical significance over clinical importance. Do not use a separate conclusion section, but instead append it as the last paragraph of the Discussion beginning something like: "In summary . . ." Take care that the conclusion is restricted to what can be justified by your experimental results. Discuss shortcomings and biases related to study design and execution.
G. References
1) The list should be numbered consecutively in the order in which they are first mentioned in the text.
In the text, citations are marked with numbers in parentheses. Every reference must be cited at least once in the text. Each reference should be cited as Arabic numerals in square brackets (e.g., [1] following the Vancouver style). If the surnames of the cited authors are given in the text, citations are indicated right after the surname. Otherwise, they are at the end of the sentence. In the former case, the last names of all authors are written for one or two authors (e.g., Han [1], Yu and Song [2]), and only the first author is recorded for three or more (e.g., Seo et al. [3]).
2) The abbreviated journal title should be used according to the NLM catalog (https://www.ncbi.nlm.nih.gov/nlmcatalog/journals) or the KoreaMed (https://www.koreamed.org/JournalBrowserNew.php ).
3) If there are six or fewer authors in a reference, list all authors. If the number of authors is greater than six, list the initial six authors, and then abbreviate the rest of the authors with by "et al."
4) Personal communications and unpublished data should be cited in the body of the paper in parentheses, not listed in the references section. Manuscripts that have been accepted for publication may be listed as "Forthcoming"; manuscripts that have been submitted or are under revision but have not been accepted should not be cited as references.
5) The use of abstracts that have not been published as full manuscripts is discouraged.
6) Authors are responsible for the accuracy and completeness of the references and text citations.
7) Number of references is limited to 35 in original articles (but, 100 in systematic reviews), 20 in case reports, and unlimited in reviews. The style and punctuation for references should follow the format illustrated in the following examples. Citing Medicine: References should follow the NLM Style Guide for Authors, Editors, and Publishers (https://www.ncbi.nlm.nih.gov/books/NBK7256/) if not specified below.
Journal article
1. Choi JW, Kim GB. Epidemiology and Clinical Outcomes of Refractory Kawasaki Disease in South Korea. Kawasaki Dis 2023;1(1):e7.
Book & Book chapter
2. Volpe JJ. Neurology of the newborn. 5th ed. Saunders; 2008.
3. Hong CE. Textbook of pediatrics. 9th ed. Korea Textbook; 2008.
4. Pan ES, Cole FS, Weinttrub PS. Viral infections of the fetus and newborn. In: Taeusch HW, Ballard RA, Gleason CA, editors. Avery’s diseases of the newborn. 8th ed. Elsevier Saunders; 2005. p. 495-529.
Website
5. International Committee of Medical Journal Editor. Uniform requirements for manuscripts submitted to biomedical journals: writing and editing for biomedical publication [Internet]. International Committee of Medical Journal Editor; c2025 [cited 2025 Apr 10]. Available from: https://www.icmje.org/about-icmje/faqs/icmje-recommendations/
H. Tables
Tables should be referenced in the main text in sequential order, and uploaded separately with the text. Each table must have a descriptive title that is composed of a phrase without a period at the top of the table. Define all abbreviations in a footnote to the table. If necessary, footnote markers can be used as listed order using superscript letters (e.g., 1), 2), 3)). Any table that has been previously published must be accompanied by the written consent of the copyright holder, and the footnote must acknowledge the original source.
I. Figures and legends
Figures must be referenced in the text in sequential order as Fig.1, Fig.2, Fig.3, and so on. Figures should clarify and augment the text. Each figure should be submitted on a separate page. Legends should be typed in 12-point font, double-spaced, and located after the references with the titles in clauses. Figures can be submitted in common formats such as JPG/JPEG/TIFF/PPT/PDF format. Photographs must be submitted electronically according to the following specifications: color photographs should be saved as TIF files in RGB at a minimum of 12.5 cm (5 in.) in width at 300 dpi; black and white photographs should be saved as TIF files in grayscale at a minimum of 12.5 cm (5 in.) in width at 300 dpi. Authors should note that all photographs with discriminability of the patients’ identity (not limited to the face) must be submitted with the publication of patient information form and the authors take the legal responsibility in case of lawsuits. Regardless of the presence of identifiable features, any clinical photograph of a patient must be accompanied by a signed informed consent form. Symbols, arrows or letters used to identify parts of the figures must be explained clearly in the legend. The illustrations of pathological tissue should state clearly the type of stain and the magnifying power (e.g., H&E, ×400), and the main contents should be marked by signs or arrows on the picture. Line drawings should be original copies. A previously published figure should be accompanied by the footnote acknowledging the original source and the consent of copyright holder.
J. Supplementary material
Supplementary material refers to files related to a specific article, which authors supply for publication alongside their article. They should generally be additional pieces to the article that could not be included in the issue or print version, such as appendices, tables, and video content. All supplementary materials will be available online alongside the full-text article. A listing of supplementary materials must be submitted at the end of the manuscript file and must be cited consecutively in the text of the submitted manuscript.
K. Corrections
To correct errors in published articles, the corresponding author should contact the journal’s editorial office with a detailed description of the proposed correction. Corrections that profoundly affect the interpretation or conclusions of the article will be reviewed by the editors. Corrections will be published as author correction or publisher correction in a later issue of the journal. Minor errors will be corrected directly in the online version of the article. An indication of the correction, along with the date it was made, will be added to the article information in both the HTML and PDF versions. A separate correction note will not be published.
6. Submission and Peer Review
A. Submission
Authors are requested to submit their papers electronically using the online manuscript submission and peer review system (https://submission.e-kd.org/). Under this online system, only corresponding authors and first authors can submit manuscripts. The process of reviewing and editing will be conducted entirely through this system. Authors and reviewers may check the progress of reviews and related questions/answers on this system. The corresponding authors will be notified of all progress in the review process. For detailed information regarding the submission, please refer to the Editorial Workflow on our website (https://www.e-kd.org/author/editorial_workflow).
B. Peer review policy
All papers, including those invited by the editor, are subject to a rigorous peer review process. KD has adopted a double-blind peer review policy, in which the identities of both the authors and reviewers are kept anonymous to each other throughout the review process. However, the editor managing the review process will have visibility of the authors' and reviewers’ identities. The Editorial Board selects reviewers based on expertise, publication history, and past reviews. During the peer review process, reviewers can interact directly or exchange information (e.g., via submission systems or email) with only an editor, which is known as “independent review.” No information about the review process or editorial decision process is published on the article page. For detailed information regarding the review process, please refer to the Editorial Workflow available on our website ().








